Myeloma News
The genomic landscape of Vk*MYC myeloma highlights shared pathways of transformation between mice and humans
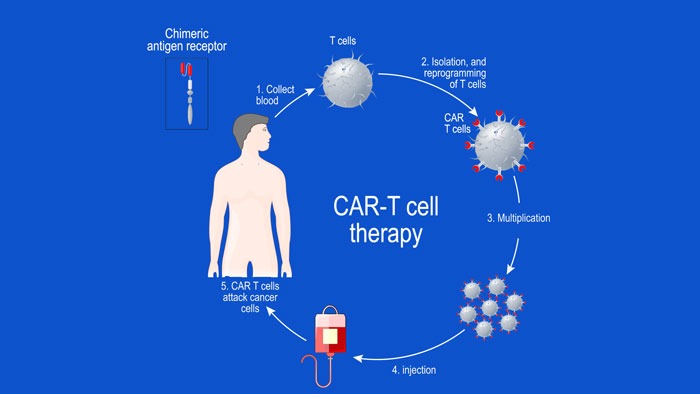
FCRL5-Directed CAR T-Cell Therapy: A Novel Therapeutic Approach for Multiple Myeloma
Source: Pharmacy Times articles
CAR T-cells specifically targeting FCRL5, when integrated with IL-15, may improve cytotoxicity, thereby enhancing the therapeutic potency of these cells.
Read More
Expert: Pharmacists Have Vital Role in Multiple Myeloma Treatment
Source: Pharmacy Times articles
The vice president of pharmacy operations at American Oncology Network says that pharmacists can contribute by educating patients and offering resources for support and treatment adherence.
Read More
Targeting myeloma essential genes using NOT Gated CAR T-cells, a computational approach
Elotuzumab Efficacy May Suffer if Given After Daratumumab in Multiple Myeloma
Source: Myeloma – Hematology Advisor
Overall survival (OS) and other efficacy outcomes were worse among patients with multiple myeloma (MM) treated with elotuzumab-based regimens after having received daratumumab compared with patients who did not have prior daratumumab, according to a retrospective study published in the Annals of Hematology. However, this decrease in efficacy was somewhat attenuated when elotuzumab was administered 180 days or more after daratumumab or if given nonsequentially.
The multicenter study analyzed data from 127 patients with MM who received elotuzumab from the Kansai Myeloma Forum registry. The primary endpoint was OS and secondary endpoints included time to next treatment (TTNT) and best response.
The median age at diagnosis was 67 and the median age at elotuzumab use was 71. The median number of prior therapies in the daratumumab cohort was 6 (range, 2-13) and was 5 (range, 1-13) in the no daratumumab cohort. Nearly all patients had prior lenalidomide, and 68% and 74% in the cohorts with or without daratumumab, respectively, had prior pomalidomide exposure.
OS was significantly longer in the no-daratumumab cohort, with a 1-year rate of 82.1% compared with 68.7% in the daratumumab cohort (P =.027).TTNT was also longer in the group without prior daratumumab at 188 days compared with 112 days in the daratumumab group (P =.024).
Best response rates were higher in the no-daratumumab group compared with the daratumumab group (P <.01), including the rates of complete response (11.3% vs 2.1%), very good partial response (11.3% vs 4.3%), and partial remission (27.5% vs 12.8%).
Among patients who were treated with daratumumab before receiving elotuzumab, nonsequential use was associated with better outcomes. The 1-year OS was 78.8% in the nonsequential group compared with 50.0% in the sequential group (P =.023). The median TTNT was longer in the nonsequential cohort at 217 days compared with 42 days in the sequential cohort (P =.031).
Best response rates were also higher with nonsequential vs sequential use for very good partial response (3.0% vs 0%) and partial remission (30.3% vs 14.3%), but this was not statistically significant (P =.120).
A longer interval of 180 or more days between the use of daratumumab and elotuzumab was associated with better outcomes than a shorter duration. The 1-year OS was 80.0% in the long interval group compared with 62.5% in the short interval group, but this did not reach statistical significance (P =.084). The mediant TTNT was 204 days and 90 days in the long and short interval groups, respectively (P =.012).
“We demonstrated that OS, TTNT, and best response were worse in the cohort treated with elotuzumab if daratumumab had been given beforehand,” the authors wrote in their report.
Disclosures: Some of the study authors declared affiliations with biotech, pharmaceutical, or device companies. Please see the original reference for a full list of disclosures.
Nurse Insights for Managing CRS in Patients With RRMM Receiving Teclistamab
Source: Myeloma – Hematology Advisor
As frontline caregivers, nurses are instrumental in promptly identifying and managing CRS in patients undergoing therapy with teclistamab. By leveraging insights from both the MajesTEC-1 trial and real-world nursing practice, nurses can enhance their ability to diagnose, monitor, and manage CRS effectively.
Cytokine release syndrome (CRS), a systemic inflammatory response commonly observed with T-cell redirecting therapies, presents challenges in patient management. Drawing from the experience of the MajesTEC-1 clinical trial (ClinicalTrials.gov Identifier: NCT04557098) and real-life nursing practice, essential guidance for nurses involved in the care of patients receiving teclistamab for relapse/refractory multiple myeloma (RRMM) was published in Seminars in Oncology Nursing.
Teclistamab, a bispecific antibody, targets CD3 on the surface of T cells and B-cell maturation antigen (BCMA) on the surface of MM cells. It is the first BCMA×CD3 bispecific antibody approved for the treatment of RRMM exposed to an immunomodulatory drug, a proteasome inhibitor, and an anti-CD38 antibody (triple-class exposed).
Despite a high incidence of CRS among patients in the trial, the majority of cases were low grade, resolved without treatment discontinuation, and aligned with supportive measures employed in real-life settings.
Insights were drawn from the experience of the MajesTEC-1 clinical trial, a phase 1/2 study of teclistamab in heavily pretreated patients with RRMM and from real-life nursing practice.
In MajesTEC-1, patients were carefully monitored for early signs and symptoms of CRS to mitigate the risk of high-grade CRS. Additionally, a survey of nurses from several study sites provided further insights into nursing best practices for CRS management from academic institutions across 3 countries.
In total, 72% of patients treated with teclistamab experienced CRS, predominantly low grade. CRS was managed with tocilizumab, intravenous fluids, low-flow oxygen therapy, and/or steroids. Notably, all cases resolved without treatment discontinuation.
“Key takeaways include the importance of recognizing and excluding infection as a differential diagnosis to ensure CRS is properly diagnosed and treated, as well as the benefits of partnering with patients and physicians to recognize and report CRS symptoms early, which is crucial for timely intervention,” wrote the authors.
In summary, this review underscores the pivotal role of nurses in recognizing and managing CRS in patients receiving teclistamab. By integrating insights from clinical trials and real-world practice, nurses can optimize patient care and outcomes in the challenging landscape of CRS management.
Disclaimer: This research was supported by Janssen Scientific Affairs, LLC. Some study authors declared affiliations with biotech, pharmaceutical, and/or device companies. Please see the original article for the full list of disclosures.
Clinical Benefits, But Not Improved ORR, Seen With KPd in Lenalidomide-Refractory Multiple Myeloma
Source: Myeloma – Hematology Advisor
Carfilzomib plus pomalidomide and dexamethasone (KPd) failed to meet the primary endpoint of overall response rate (ORR) among patients with early relapsed/refractory multiple myeloma (RRMM) who were lenalidomide-refractory, according to the results of the phase 2 SELECT trial published in the journal Leukemia & Lymphoma. However, the researchers observed other clinical benefits with KPd, including progression-free survival (PFS) and overall survival (OS).
In the open-label SELECT study (ClinicalTrials.gov Identifier: NCT04191616), researchers treated 52 patients with RRMM who had received 1-2 prior lines of therapy with KPd. The primary endpoint was ORR and secondary endpoints included minimal residual disease (MRD) negativity (10-5) at 12 months, duration of response (DOR), time to response (TTR), PFS, and OS.
At baseline, the median age was 68 years, 54% of patients were female, 90% were White, and 8% were Hispanic/Latino. There were 38% of patients with high-risk cytogenetics, and 58% had a prior transplant. All patients were refractory to lenalidomide and 37% were triple-refractory.
There were 58% of patients who demonstrated an overall response, including 6% with a complete response and 35% with a very good partial response. There were 3.8% of patients who achieved MRD negativity at 12 months and 9.6% who achieved it at any time. The median duration of response was 20.3 months and the median TTR was 1.0 months.
During a median follow-up of 15.5 months, the median PFS was 11.1 months and the 1- and 2-year PFS rates were 47% and 33%, respectively. The median OS was 18.8 months during a median follow-up of 16.6 months. The 1- and 2-year OS was 59% and 47%, respectively.
Grade 3 or higher treatment-emergent adverse events (TEAEs) occurred in 67% of patients, with the most common being hematologic. The most common nonhematologic grade 3 or higher TEAEs included acute kidney injury, rash, asthenia, and respiratory failure. There were no treatment-related deaths.
“KPd may be considered an attractive treatment option for an increasing number of patients with relapsed and multidrug-resistant MM,” the study authors wrote in their report. “With lenalidomide as [a standard of care] and increasing use of anti-CD38 mAbs in the frontline setting, effective treatment combinations for lenalidomide-refractory and CD38-refractory patients may be of increasing importance.”
Disclosures: This study was supported by Amgen. Please see the original reference for a full list of disclosures.
Single-cell multiomic dissection of response and resistance to chimeric antigen receptor T cells against BCMA in relapsed multiple myeloma
Daratumumab-based quadruplet therapy for transplant-eligible newly diagnosed multiple myeloma with high cytogenetic risk
Bispecific CAR T cell therapy targeting BCMA and CD19 in relapsed/refractory multiple myeloma: a phase I/II trial
Recent Publications
Isatuximab plus pomalidomide and dexamethasone in patients with relapsed/refractory multiple myeloma in real-world: The retrospective IMAGE study
Eur J Haematol. 2024 May 7. doi: 10.1111/ejh.14225. Online ahead of print. ABSTRACT BACKGROUND: IMAGE is a retrospective cohort study of patients enrolled in early access programs (EAPs) in France with relapsed/refractory multiple myeloma (RRMM) receiving isatuximab with pomalidomide and...
KIF22 promotes multiple myeloma progression by regulating the CDC25C/CDK1/cyclinB1 pathway
J Cancer Res Clin Oncol. 2024 May 7;150(5):239. doi: 10.1007/s00432-024-05747-w. ABSTRACT PURPOSE: Multiple myeloma (MM) is an incurable hematological malignancy characterized by clonal proliferation of malignant plasma B cells in bone marrow, and its pathogenesis remains unknown. The aim of this...
Human IL-6 fosters long-term engraftment of patient derived disease-driving myeloma cells in immunodeficient mice
JCI Insight. 2024 May 7:e177300. doi: 10.1172/jci.insight.177300. Online ahead of print. ABSTRACT Multiple myeloma is a largely incurable and life-threatening malignancy of antibody-secreting plasma cells. An effective and widely available animal model that recapitulates human myeloma and related...
FDA Approval Summary: Ciltacabtagene Autoleucel for Relapsed or Refractory Multiple Myeloma
Clin Cancer Res. 2024 May 7. doi: 10.1158/1078-0432.CCR-24-0378. Online ahead of print. ABSTRACT In February 2022, the U.S. Food and Drug Administration approved ciltacabtagene autoleucel, a chimeric antigen receptor (CAR) T cell therapy targeting the B-cell maturation antigen (BCMA), for adult...
Mass Spectrometry-Based Assessment of M-protein in Peripheral Blood During Maintenance Therapy in Multiple Myeloma
Blood. 2024 May 7:blood.2024024041. doi: 10.1182/blood.2024024041. Online ahead of print. ABSTRACT Mass spectrometry (MS) can detect multiple myeloma-derived monoclonal proteins in peripheral blood (PB) with high sensitivity, potentially serving as a PB assay for measurable residual disease (MRD)....
Understanding health outcome preferences of older adults diagnosed with multiple myeloma
J Geriatr Oncol. 2024 May 6;15(5):101781. doi: 10.1016/j.jgo.2024.101781. Online ahead of print. ABSTRACT INTRODUCTION: Health outcome preferences of older adults with cancer vary based on burden/intensity of treatment and its impact on health outcomes such as survival, quality of life, and...
The genomic landscape of Vk*MYC myeloma highlights shared pathways of transformation between mice and humans
Nat Commun. 2024 May 7;15(1):3844. doi: 10.1038/s41467-024-48091-w. ABSTRACT Multiple myeloma (MM) is a heterogeneous disease characterized by frequent MYC translocations. Sporadic MYC activation in the germinal center of genetically engineered Vk*MYC mice is sufficient to induce plasma cell...
CAR T cells in multiple myeloma: lessons learned
Nat Rev Clin Oncol. 2024 May 7. doi: 10.1038/s41571-024-00898-8. Online ahead of print. NO ABSTRACT PMID:38714789 | DOI:10.1038/s41571-024-00898-8
Expert Recommendations & Patient Perspectives for Treating Relapsed/Refractory Multiple Myeloma
In this webcast, expert faculty discuss best practices in treating patients with relapsed/refractory multiple myeloma and discuss the patient's perspective on treatment options. Learners will also get a look ahead at emerging therapeutic strategies and clinical trials...
Overview of Key Early-phase CAR T-cell Therapy Studies in Relapsed or Refractory Multiple Myeloma
In this article, we look at some of the early-phase clinical trials, where chimeric antigen receptor (CAR) T-cell therapy has been targeted to the B-cell maturation antigen (BCMA) in patients with relapsed or refractory MM (RRMM). The anti-BCMA CAR T-cell...
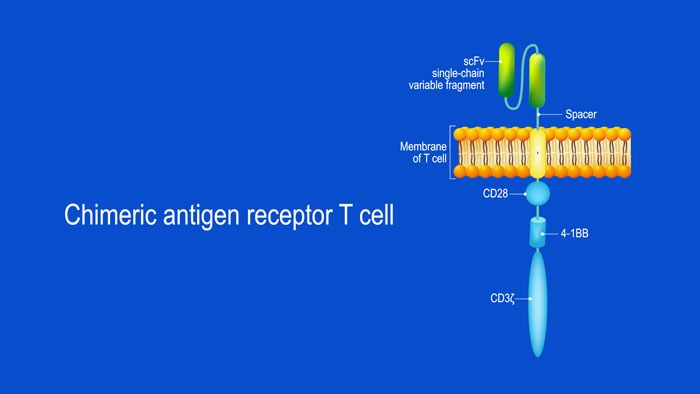
What Are Chimeric Antigen Receptor (CAR) T-cells and What is Their Potential Target Site in Multiple Myeloma?
CAR T-cell structure CARs are typically composed of four regions: (1) an extracellular antigen-binding domain; (2) a hinge or spacer peptide; (3) a transmembrane domain that anchors the CAR to the cell membrane; and (4) one or more intracellular signalling domains...